Adenocarcinoma is a relatively rare malignancy in the urinary bladder, which can originate primarily within the bladder or secondarily from various other organs. Alfa Cytology, as a leading global authority in bladder cancer research, leverages our extensive expertise and advanced platform to provide unparalleled research services for adenocarcinoma.
Introduction to Adenocarcinoma
Grossly, bladder adenocarcinoma typically originates from the trigone and posterior wall but can manifest in any region of the bladder. It commonly presents as a solitary lesion, in contrast to urothelial carcinoma which tends to be multifocal. Macroscopically, the tumor may exhibit papillary, sessile, solid, or ulcerating characteristics. The cut surface often displays a gelatinous appearance due to abundant mucin production.
Histologically, bladder adenocarcinoma exhibits various growth patterns.
- Enteric (colonic or intestinal) - The enteric pattern is composed of intestinal-type glands with pseudostratified columnar cells and nuclear atypia, closely resembling colorectal adenocarcinoma. It may produce intracellular or extracellular mucin, and necrosis is not infrequent.
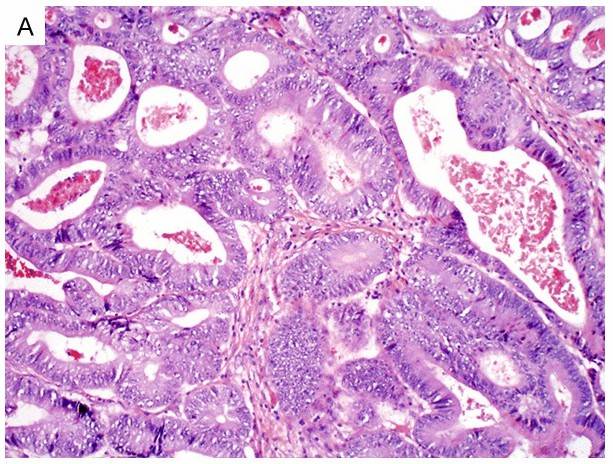 Fig.1 Enteric-type bladder adenocarcinoma shows intestinal-type glands with columnar cells and marked cytologic atypia. (Dadhania, V., et al., 2015)
Fig.1 Enteric-type bladder adenocarcinoma shows intestinal-type glands with columnar cells and marked cytologic atypia. (Dadhania, V., et al., 2015)
- Mucinous (colloid) - The mucinous pattern produces abundant extracellular mucin with tumor cells floating in a pool of mucin.
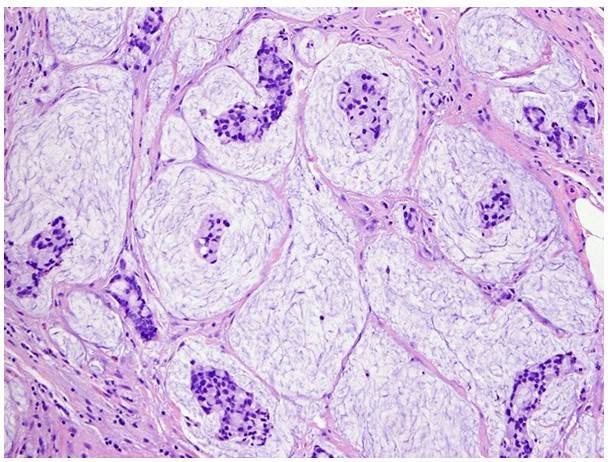 Fig.2 Mucinous-type bladder adenocarcinoma produces abundant extracellular mucin, forming a pool of mucin with floating tumor cells. (Dadhania, V., et al., 2015)
Fig.2 Mucinous-type bladder adenocarcinoma produces abundant extracellular mucin, forming a pool of mucin with floating tumor cells. (Dadhania, V., et al., 2015)
- Signet ring cell - The signet ring cell pattern is composed of diffusely infiltrative poorly differentiated cells with prominent intracellular mucin and indented eccentric nuclei. These tumors tend to present at an advanced stage and carry a worse prognosis than other variants.
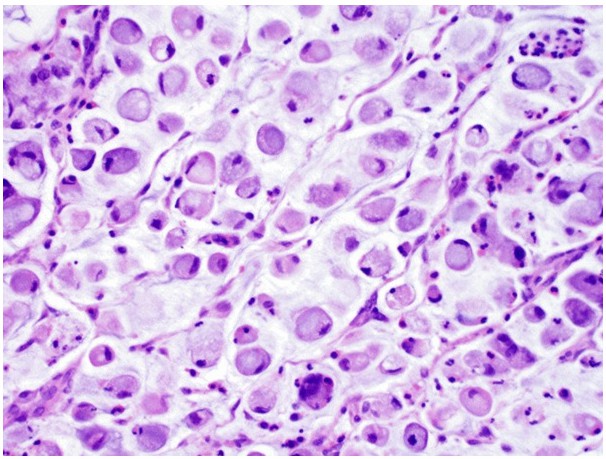 Fig.3 Signet ring cell-type bladder adenocarcinoma is characterized by large intracellular mucin vacuoles that displace nuclei to the periphery. (Dadhania, V., et al., 2015)
Fig.3 Signet ring cell-type bladder adenocarcinoma is characterized by large intracellular mucin vacuoles that displace nuclei to the periphery. (Dadhania, V., et al., 2015)
- Not otherwise specified (NOS) - The NOS type has non-specific glandular growth.
- Mixed patterns - Tumors with more than one pattern are classified as the mixed type.
Therapy Development for Adenocarcinoma
The following is some therapies information regarding adenocarcinoma of the bladder cancer.
|
Therapeutics |
Mechanism of Action |
Phase |
Observed Effects |
| Nivolumab |
PD-1 Inhibitor |
Phase II |
Increased Overall Survival Rates |
| Atezolizumab |
PD-L1 Inhibitor |
Phase III |
Improved Response Rates in Metastatic Cases |
| Pembrolizumab |
PD-1 Inhibitor |
Phase II |
Effective in Treating High-Risk, BCG-Unresponsive Patients |
| Enfortumab Vedotin |
ADC Targeting Nectin-4 |
Phase III |
Significant Tumor Reduction and Improved Patient Outcomes |
| Erdafitinib |
FGFR Inhibitor |
Phase II |
Efficacy in Patients with FGFR Genetic Alterations |
Our Services
Alfa Cytology offers a comprehensive range of research services tailored to address the complex pathophysiology of adenocarcinoma and facilitate the development of effective diagnostic and therapeutic strategies.
Case Study - RT-112 and Gemcitabine-Resistant RT-112 Bladder Cancer CDX Model
Model Introduction
The RT-112 and gemcitabine-resistant RT-112 bladder cancer cell line-derived xenograft (CDX) model provides a standardized preclinical platform for evaluating the efficacy of the fibroblast growth factor receptor (FGFR) inhibitor. This model utilizes the RT-112 human bladder cancer cell line, which harbors FGFR3 alterations, to study targeted inhibition of FGFR signaling and its antitumor effects.
Model Information
- Model: RT-112 Bladder Cancer CDX Model and Gemcitabine-Resistant RT-112 Bladder Cancer CDX Model
- Animal: BALB/c Nude Mice
- Weight: 18-20 g
Model Construction
RT-112 cells or gemcitabine-resistant RT-112 cells were subcutaneously inoculated into the flanks of BALB/c nude mice. When tumor volumes reached approximately 150-200 mm3, mice were randomized into treatment groups.
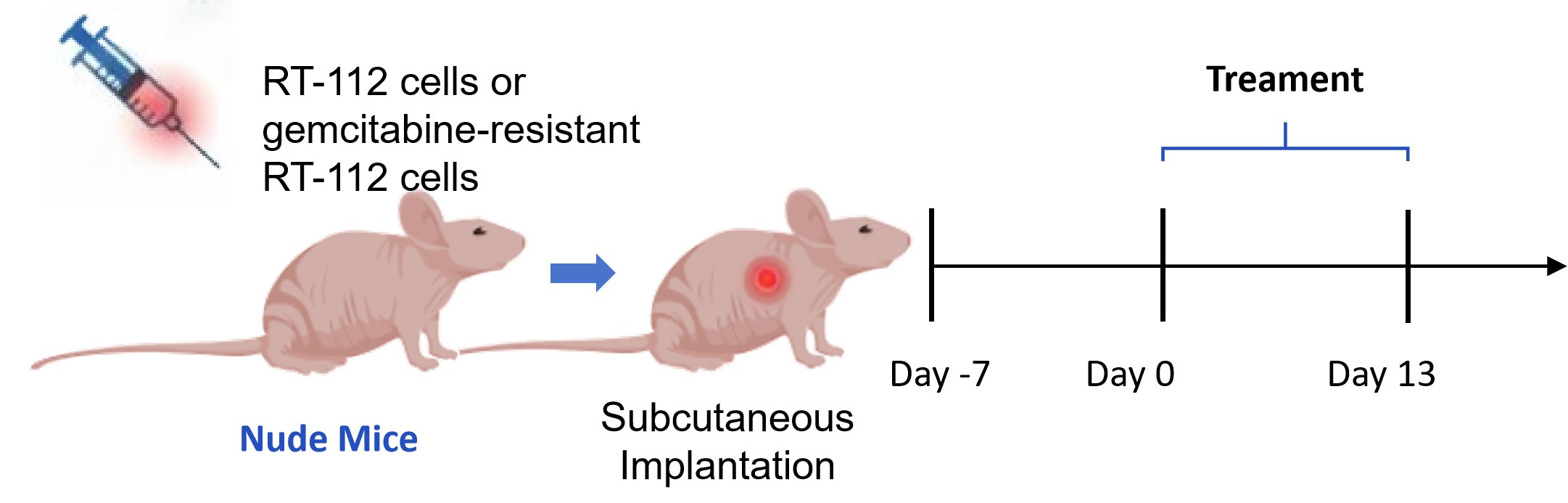 Fig. 4 Workflow of the RT-112 CDX model establishment and drug A treatment regimen. (Source: Alfa Cytology)
Fig. 4 Workflow of the RT-112 CDX model establishment and drug A treatment regimen. (Source: Alfa Cytology)
In Vivo Efficacy Evaluation
This study employed the RT-112 CDX model and gemcitabine-resistant RT-112 CDX model to evaluate the antitumor efficacy of drug A.
- Drug A Efficacy in Standard RT-112 Model: Oral administration of drug A (3 mg/kg) resulted in significant tumor growth inhibition and induced marked tumor regression in the RT-112 CDX model, demonstrating potent antitumor activity against FGFR3-altered bladder cancer.
- Drug A Efficacy in Gemcitabine-Resistant RT-112 Model: Drug A (3 mg/kg) maintained significant antitumor efficacy in the gemcitabine-resistant RT-112 model, confirming its retained activity against FGFR3-driven tumors with acquired chemotherapy resistance.
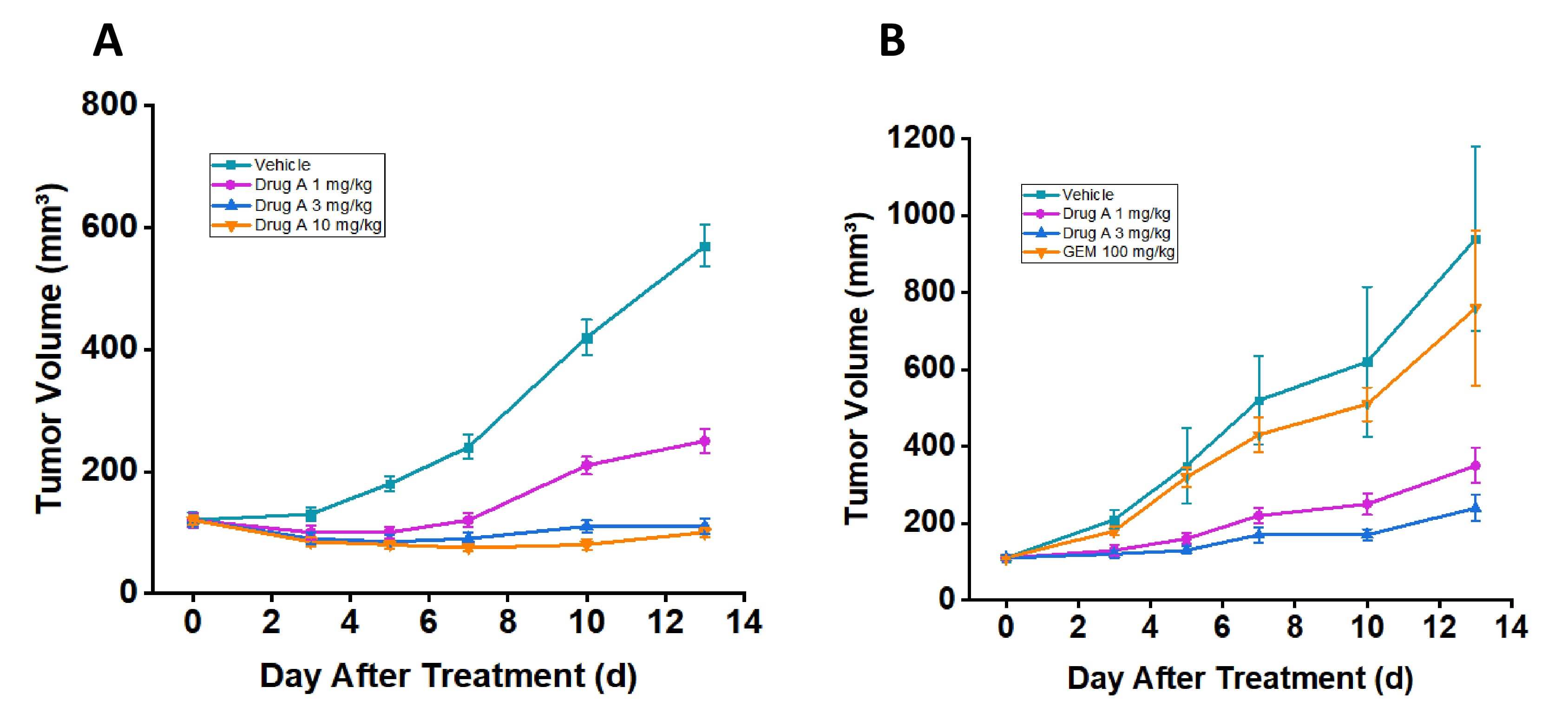 Fig. 5 Drug A demonstrates potent anti-tumor activity in both RT-112 and gemcitabine-resistant RT-112 CDX models. (A) Tumor volume in the RT-112 model. (B) Tumor volume in gemcitabine-resistant RT-112 model. Data are presented as mean ± SEM. (Source: Alfa Cytology)
Fig. 5 Drug A demonstrates potent anti-tumor activity in both RT-112 and gemcitabine-resistant RT-112 CDX models. (A) Tumor volume in the RT-112 model. (B) Tumor volume in gemcitabine-resistant RT-112 model. Data are presented as mean ± SEM. (Source: Alfa Cytology)
Contact Us
Ongoing research and advancements in the understanding of adenocarcinoma biology continue to drive the development of innovative therapeutic approaches. At Alfa Cytology, we are dedicated to providing comprehensive and state-of-the-art solutions for adenocarcinoma therapy development. For further information regarding our extensive services, please do not hesitate to contact us.
Reference
- Dadhania, V., Czerniak, B., & Guo, C. C. Adenocarcinoma of the urinary bladder. American journal of clinical and experimental urology, 2015, 3(2), 51–63.
For research use only. Not intended for any clinical use.
Related Services

 Fig.1 Enteric-type bladder adenocarcinoma shows intestinal-type glands with columnar cells and marked cytologic atypia. (Dadhania, V., et al., 2015)
Fig.1 Enteric-type bladder adenocarcinoma shows intestinal-type glands with columnar cells and marked cytologic atypia. (Dadhania, V., et al., 2015) Fig.2 Mucinous-type bladder adenocarcinoma produces abundant extracellular mucin, forming a pool of mucin with floating tumor cells. (Dadhania, V., et al., 2015)
Fig.2 Mucinous-type bladder adenocarcinoma produces abundant extracellular mucin, forming a pool of mucin with floating tumor cells. (Dadhania, V., et al., 2015) Fig.3 Signet ring cell-type bladder adenocarcinoma is characterized by large intracellular mucin vacuoles that displace nuclei to the periphery. (Dadhania, V., et al., 2015)
Fig.3 Signet ring cell-type bladder adenocarcinoma is characterized by large intracellular mucin vacuoles that displace nuclei to the periphery. (Dadhania, V., et al., 2015)


 Fig. 4 Workflow of the RT-112 CDX model establishment and drug A treatment regimen. (Source: Alfa Cytology)
Fig. 4 Workflow of the RT-112 CDX model establishment and drug A treatment regimen. (Source: Alfa Cytology)  Fig. 5 Drug A demonstrates potent anti-tumor activity in both RT-112 and gemcitabine-resistant RT-112 CDX models. (A) Tumor volume in the RT-112 model. (B) Tumor volume in gemcitabine-resistant RT-112 model. Data are presented as mean ± SEM. (Source: Alfa Cytology)
Fig. 5 Drug A demonstrates potent anti-tumor activity in both RT-112 and gemcitabine-resistant RT-112 CDX models. (A) Tumor volume in the RT-112 model. (B) Tumor volume in gemcitabine-resistant RT-112 model. Data are presented as mean ± SEM. (Source: Alfa Cytology)