Non-muscle Invasive Bladder Cancer
The majority of bladder cancer cases (75%) are classified as non-muscle invasive bladder cancer (NMIBC), which refers to cancer that is limited to the mucosa and submucosa of the bladder. Alfa Cytology offers comprehensive research services aimed at supporting the development of diagnostics and therapeutics for NMIBC, leveraging our expertise in preclinical research to expedite your breakthroughs.
Overview of Non-muscle Invasive Bladder Cancer
The category of non-muscle invasive bladder cancer encompasses papillary tumors located within the mucosa (stage Ta), tumors that invade the lamina propria (stage T1), and flat high-grade lesions known as carcinoma in situ (CIS). NMIBC is characterized by its limited invasion, rendering it less aggressive compared to muscle-invasive bladder cancer. In contrast to muscle-invasive cancer, NMIBC is primarily managed through local endoscopic/intravesical therapy and surveillance.
Definitions of high-risk non-muscle-invasive bladder cancer according to the American and European Urologic Associations.
 European Urologic Association
European Urologic Association
- Carcinoma in situ
- High-grade T1 tumors
- Recurrent or multifocal or large (> 3 cm) high-grade Ta tumors
- Any tumor following BCG failure
- Lymphovascular invasion or non-urothelial histology
- High-grade tumor involving prostatic urethra
 American Urological Association
American Urological Association
- Carcinoma in situ
- Any high-grade tumor
- Any T1 tumor
- Multiple, recurrent, and large low-grade Ta tumors
Highest risk:
- T1 HG with CIS
- Multiple, large, or recurrent T1 high-grade tumors
- T1 with CIS in prostatic urethra
- Some variant histology or lymphovascular invasion
Therapy Development for NMIBC
The recent advancements in the therapy of NMIBC have introduced several novel therapeutic approaches, with a focus on enhancing efficacy and reducing disease recurrence. Here are some noteworthy new therapies.
|
Therapy |
Therapeutics |
Phase |
Notes |
 |
Immunotherapy |
PD-1/PD-L1 Inhibitors |
Clinical Trials/Approved |
Examples include Atezolizumab, Pembrolizumab |
| BCG (Bacillus Calmette-Guerin) |
Standard Treatment/Resistance Issues |
Ongoing research on alternatives due to resistance development |
 |
Targeted Therapy |
Small Molecule Inhibitors |
In Development |
Targets include FGFR3, HER2 |
 |
Cancer Vaccines |
Cancer Vaccines |
Early Research/Clinical Trials |
Exploring immune system activation against cancer cells |
 |
New Drug Types |
Novel Chemotherapy Drugs |
In Development |
Includes nanomedicines and new chemotherapeutic agents |
| Antibody-Drug Conjugates (ADC) |
In Development |
Combines antibodies and cytotoxins targeting specific NMIBC markers |
Our Services
At Alfa Cytology, we offer a comprehensive suite of services tailored to the unique needs of NMIBC research. Our preclinical expertise and state-of-the-art facilities ensure that your projects receive the highest quality support from discovery through to development.
Case Study - Orthotopic Murine Non-Muscle-Invasive Bladder Cancer (NMIBC) Model
Model Introduction
The NMIBC model provides a clinically relevant platform for evaluating novel intravesical immunotherapies. This model recapitulates the local tumor microenvironment and allows direct assessment of xenogeneic urothelial cell therapy alone or in combination with chemotherapy or immune checkpoint inhibitors, enabling the study of immune-mediated antitumor responses and potential synergistic effects.
Model Information
- Model: Orthotopic MB49-Luc NMIBC Model
- Animal: C57BL/6 Mice
- Weight: 18-22 g
Model Construction
Orthotopic NMIBC models were established by intravesical instillation of MB49-Luc cells into C57BL/6 mouse bladders. Tumor growth was monitored via bioluminescence imaging.
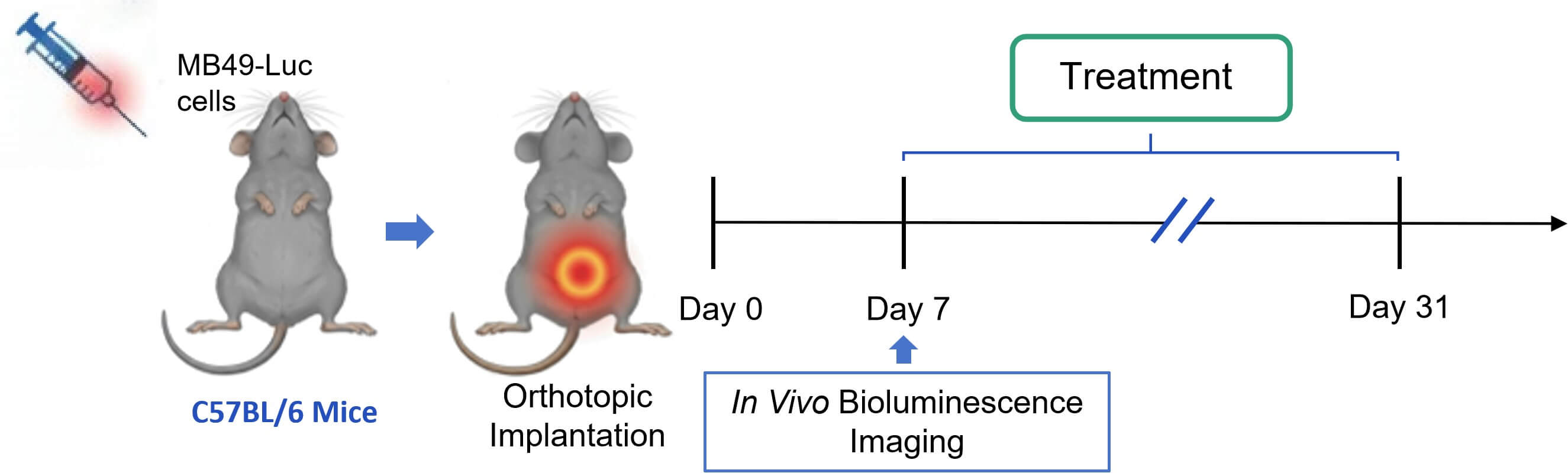 Fig. 2 Workflow of MB49-Luc NMIBC mouse model establishment and combination therapy regimen. (Source: Alfa Cytology)
Fig. 2 Workflow of MB49-Luc NMIBC mouse model establishment and combination therapy regimen. (Source: Alfa Cytology)
In Vivo Efficacy Evaluation
This established orthotopic NMIBC model was employed to systematically evaluate the anti-tumor efficacy and immunomodulatory effects of intravesical cell therapy alone and in combination with chemotherapy (gemcitabine, GEM) or immune checkpoint inhibition (anti-PD-1).
- Cell Monotherapy: Intravesical cell alone demonstrated significant tumor growth inhibition and prolonged survival compared to the vehicle control.
- GEM or Anti-PD-1 Monotherapy: Both monotherapies showed variable but significant anti-tumor effects compared to the control.
- Combination Therapies: The combination of cell therapy with either GEM or anti-PD-1 resulted in superior tumor growth inhibition, greater reduction in final bladder tumor weight.
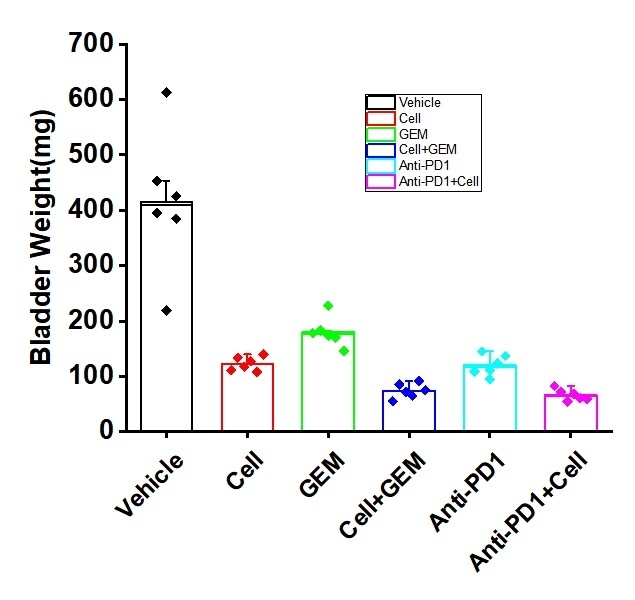 Fig. 3 Anti-tumor efficacy of cell therapy in the orthotopic NMIBC model. Data are presented as mean ± standard error (SEM). (Source: Alfa Cytology)
Fig. 3 Anti-tumor efficacy of cell therapy in the orthotopic NMIBC model. Data are presented as mean ± standard error (SEM). (Source: Alfa Cytology)
Contact Us
By partnering with Alfa Cytology, you gain access to cutting-edge technologies and expert guidance, driving your NMIBC research forward with confidence. Contact us today to learn more about how we can support your journey from concept to preclinical success.
For research use only. Not intended for any clinical use.
Related Services





 Fig. 2 Workflow of MB49-Luc NMIBC mouse model establishment and combination therapy regimen. (Source: Alfa Cytology)
Fig. 2 Workflow of MB49-Luc NMIBC mouse model establishment and combination therapy regimen. (Source: Alfa Cytology)  Fig. 3 Anti-tumor efficacy of cell therapy in the orthotopic NMIBC model. Data are presented as mean ± standard error (SEM). (Source: Alfa Cytology)
Fig. 3 Anti-tumor efficacy of cell therapy in the orthotopic NMIBC model. Data are presented as mean ± standard error (SEM). (Source: Alfa Cytology)