Small Cell Carcinoma of the Bladder
Small cell carcinoma of the bladder (SCCB) is an uncommon and highly aggressive variant of bladder cancer, comprising less than 1% of all bladder cancer cases. Alfa Cytology provides specialized research services to facilitate the investigation and advancement of treatments for SCCB.
Introduction to Small Cell Carcinoma of the Bladder
The histological features of small cell carcinoma of the bladder resemble those of small cell lung cancer, including the presence of neuroendocrine cells. This aggressive malignancy is characterized by small, round, and densely packed cells, leading to rapid growth and early metastasis. Due to its highly aggressive nature, SCCB often presents at an advanced stage and carries a poor prognosis.
- Neuroendocrine Features
Small cell carcinoma is a high-grade neuroendocrine tumor characterized by the expression of neuroendocrine markers such as synaptophysin, chromogranin, and CD56.
- Genetic Alterations
Common mutations include those in TP53, RB1, and genes involved in the Notch signaling pathway.
- Rapid Proliferation
These tumors have a high mitotic rate and rapid growth, leading to early metastasis.
- Association with Other Histologies
Small cell carcinoma often coexists with other types of bladder cancer, such as urothelial carcinoma, indicating a shared pathogenesis or a dedifferentiation process.
Therapy Development for SCCB
In recent years, there have been advancements in the field of SCCB therapy, leading to the emergence of new and promising approaches. Some of these emerging therapeutic approaches include the follows.
|
Therapy |
Therapeutics |
Phase |
Notes |
| Chemotherapy |
Platinum-based Regimens |
Standard Treatment |
Includes Combinations like Etoposide and Cisplatin |
| Immunotherapy |
PD-1/PD-L1 Inhibitors |
Early Research/Clinical Trials |
Evaluated in Small Cell Carcinoma |
| Targeted Therapy |
PARP Inhibitors |
In Development |
Focus on Specific Genetic Alterations in Small Cell Carcinoma |
Our Services
At Alfa Cytology, we provide a comprehensive range of therapy development services specifically designed to address the unique challenges presented by SCCB and other bladder cancers.

Modeling Services
Our modeling services offer state-of-the-art techniques to accurately simulate SCCB in various experimental settings.
Case Study - Small Cell Bladder Carcinoma (SCBC) PDX Model
Model Introduction
The small cell bladder carcinoma (SCBC) patient-derived xenograft (PDX) model provides a clinically relevant and biologically faithful preclinical platform for evaluating novel targeted therapies against this aggressive neuroendocrine malignancy. This model recapitulates key molecular features of human SCBC, including high expression of the Notch ligand DLL3, enabling the study of DLL3-targeted antibody-drug conjugates (ADCs) as a promising therapeutic strategy.
Model Information
Model Details
- Model: Small Cell Bladder Carcinoma (SCBC) PDX Model
- Animal: NOD/SCID Mice
- Weight: 18-20 g
Patient Information
- Sex: Female
- Race/Ethnicity: White
- Treatment: Treatment-naïve prior to sample collection.
- Age: 67
- Diagnosis: High-grade neuroendocrine carcinoma of the bladder (Small Cell Bladder Cancer, SCBC).
Model Construction
The PDX model was established from a cystectomy specimen of a high-grade neuroendocrine bladder carcinoma. Dissociated tumor cells were subcutaneously implanted into the lower mammary fat pad of NOD/SCID mice. Tumor growth was monitored, and when average tumor volumes reached 140-200 mm3, mice were randomized into treatment groups.
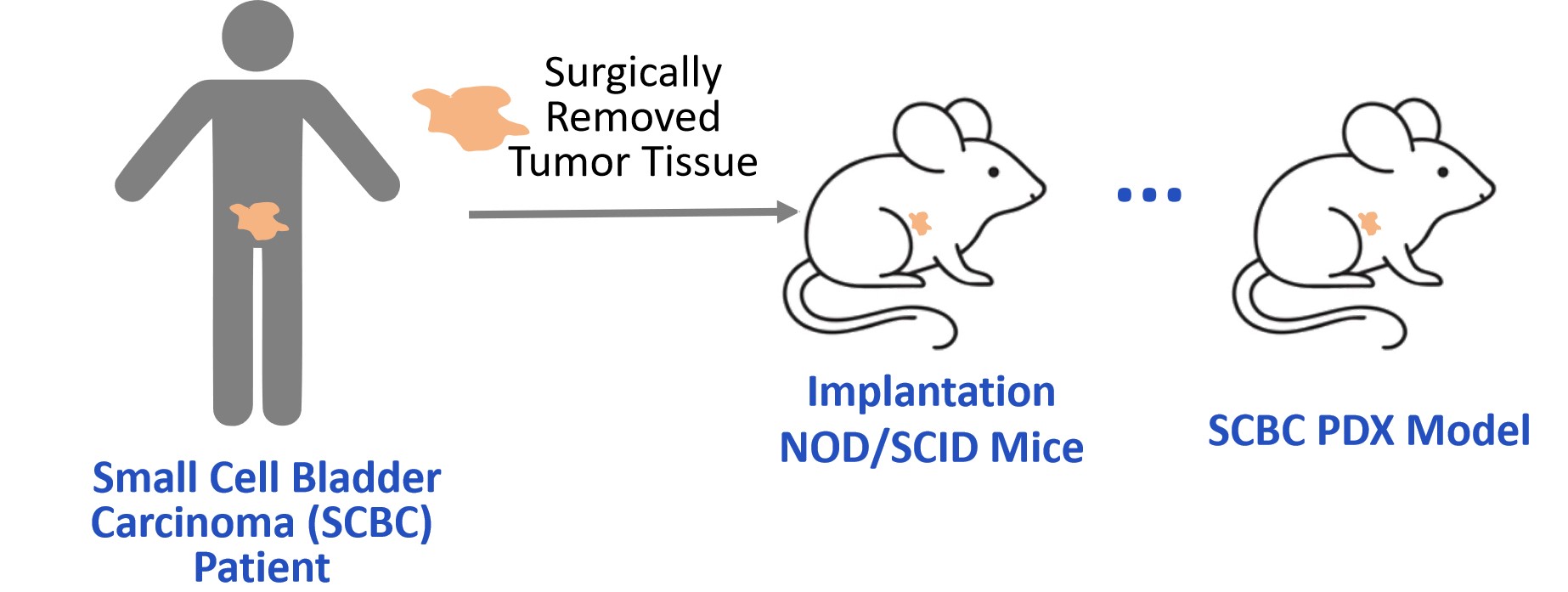 Fig. 1 Workflow of SCBC PDX model establishment and treatment regimen. (Source: Alfa Cytology)
Fig. 1 Workflow of SCBC PDX model establishment and treatment regimen. (Source: Alfa Cytology)
In Vivo Efficacy Evaluation
This study employed the established SCBC PDX model to evaluate the antitumor efficacy of the DLL3-targeting antibody-drug conjugate (ADC).
- ADC Monotherapy: A single dose of ADC demonstrated superior and durable antitumor efficacy compared to both vehicle control and standard chemotherapy (cisplatin + etoposide). ADC treatment resulted in sustained tumor suppression for >100 days, whereas chemotherapy only transiently inhibited growth.
- Target Specificity & Tumor-Initiating Cell (TIC) Depletion: ADC significantly reduced the frequency of tumor-initiating cells compared to vehicle control, indicating its ability to target the stem-like cell population responsible for tumor recurrence. An isotype control group showed only modest non-specific activity.
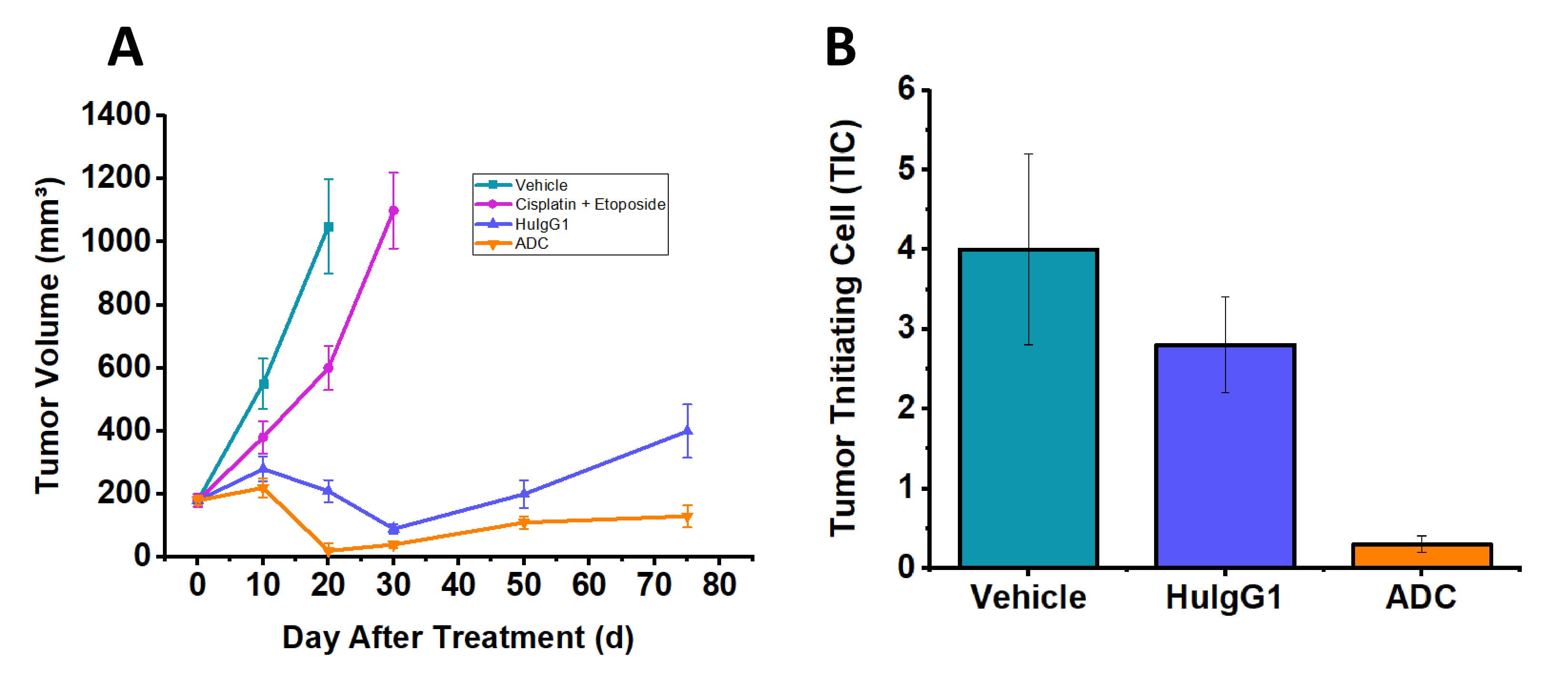 Fig. 2 ADC anti-tumor efficacy in the SCBC PDX model. (A) Tumor volume. (B) Residual tumor initiating cell (TIC) frequency. Data are presented as mean ± SEM. (Source: Alfa Cytology)
Fig. 2 ADC anti-tumor efficacy in the SCBC PDX model. (A) Tumor volume. (B) Residual tumor initiating cell (TIC) frequency. Data are presented as mean ± SEM. (Source: Alfa Cytology)
Contact Us
Alfa Cytology is dedicated to providing high-quality research and development services to support your SCCB projects. With a strong emphasis on innovation and collaboration, our goal is to expedite the discovery and advancement of effective therapies for small cell carcinoma of the bladder. Please don't hesitate to contact us today to explore how we can assist with your research requirements and contribute significantly to combating this formidable disease.
For research use only. Not intended for any clinical use.
Related Services





 Fig. 1 Workflow of SCBC PDX model establishment and treatment regimen. (Source: Alfa Cytology)
Fig. 1 Workflow of SCBC PDX model establishment and treatment regimen. (Source: Alfa Cytology)  Fig. 2 ADC anti-tumor efficacy in the SCBC PDX model. (A) Tumor volume. (B) Residual tumor initiating cell (TIC) frequency. Data are presented as mean ± SEM. (Source: Alfa Cytology)
Fig. 2 ADC anti-tumor efficacy in the SCBC PDX model. (A) Tumor volume. (B) Residual tumor initiating cell (TIC) frequency. Data are presented as mean ± SEM. (Source: Alfa Cytology)