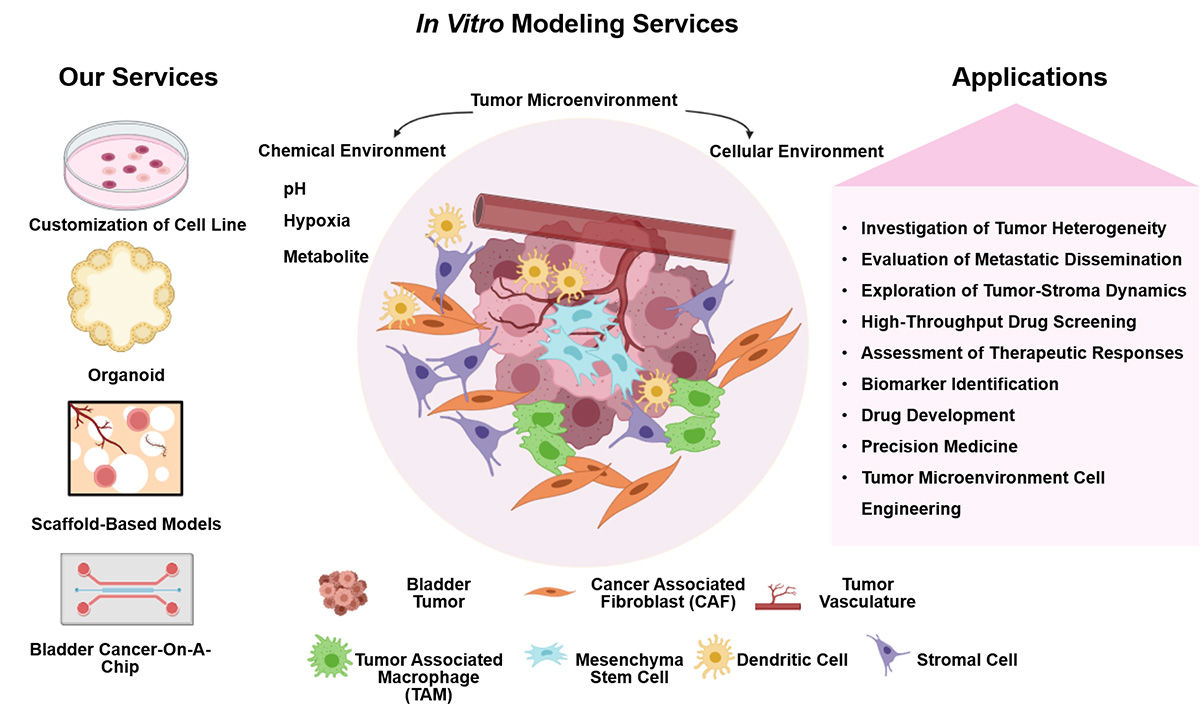
In Vitro Modeling Services for Bladder Cancer
In vitro modeling involves the cultivation and manipulation of bladder cancer cells or tissues in a controlled laboratory environment outside of living organisms. In vitro models serve as valuable tools for hypothesis testing, drug screening, and mechanistic investigations. At Alfa Cytology, we provide our clients with comprehensive in vitro modeling services for bladder cancer.
Introduction of In vitro Models for Bladder Cancer
In vitro modeling offers several advantages in bladder cancer research. Firstly, it provides a controlled and reproducible experimental setting, allowing researchers to precisely manipulate and monitor specific variables. In vitro models also offer a cost-effective alternative to animal models, reducing the reliance on animal testing and ethical concerns associated with such studies.
Advantage and deficiency of in vitro models are shown below:
|
Source |
Advantage |
Deficiency |
| In vitro 2D models |
Cell lines from human, mouse, rat and dog. |
- Easy to culture;
- Economic;
- Readily available;
- Easy to manipulate;
- Mouse and rat cell lines can be implanted into immunocompetent host to study immunotherapy;
- The most widely used model in BC research.
|
- Different genetic and epigenetic compositions from those of human cancers because of long term in vitro culture;
- Relatively pure cancer cell population
- Lack of supporting cells and TME
- Human cancer lines can only be implanted into immunocompromised mice and are not suitable to study immunotherapy
|
| In vitro 3D models |
Organoids |
- 3D structure with supporting cells and TME;
- Similar genetic alterations as the parental cancer cells;
- Relatively high concordance of drug sensitivity with parental cancers;
- Cheaper and easier than in vivo models.
|
- Lack of in vivo factors, such as blood circulation
- Lack of dynamic immune system
- Special medium with growth factor support
|
| 3D printing |
- 3D structure with supporting cells and TME;
- Precise control of cancer cells, stromal cells and stroma;
- Rapid production of large numbers of tumor 3D printings.
|
- Need for special 3D printer
- Lack of in vivo factors
- Difference of 3D structure in 3D printing compared to native cancers
- Lack of dynamic immune system as in vivo
|
| Bladder Cancer-On-A-Chip |
- In vivo relevant physiological microenvironment
- Vasculature-like perfusion
- Precise control of chemical gradient flows
- Mechanical forces
- Fully personalized system
|
- High expertise barrier
- High time-and resource-consuming
- Short-term culture
- Limitations in high-throughput analyses
- Patient availability
|
Our Services
At Alfa Cytology, we are at the forefront of providing comprehensive preclinical contract research organization (CRO) services in the field of bladder cancer, with a specific focus on in vitro model development. Some of the key services we offer include:
Case Study - The 5637 Cell Model
Model Introduction
The 5637 human bladder cancer cell line, established from a primary Grade II bladder transitional cell carcinoma, serves as a well-characterized model for studying urothelial carcinoma biology and therapeutic responses. It exhibits typical epithelial morphology, wild-type p53 status, and reliable tumorigenicity, making it a valuable tool for preclinical research on tumor progression, drug sensitivity, and novel therapeutic strategies in bladder cancer.
Model Information
- Origin: Human primary bladder transitional cell carcinoma (Grade II).
- Classification: Bladder urothelial carcinoma (transitional cell carcinoma) cell line.
- Genetic Background: Characterized by wild-type TP53 gene status.
- Morphology: Epithelial-like.
- Growth Pattern: Adherent.
- Key Markers: Expresses urothelial differentiation markers; commonly used in studies of bladder cancer proliferation and drug response.
- Cancer Type: Muscle-Invasive Bladder Cancer
Model Validation (In Vivo)
The reliable tumorigenicity of the 5637 cell line was validated in vivo by establishing a subcutaneous xenograft model. 5637 cells (5×106 or 1×107 cells/mouse) were inoculated into the flank of 7-week-old immunodeficient C-NKG mice. Longitudinal monitoring demonstrated efficient and stable tumor engraftment. Tumor volumes reached 100-200 mm3 by approximately day 7-8 post-inoculation and progressed to the study endpoint volume of 2000 mm3 by day 64. Based on this growth profile, a therapeutic intervention window is optimally initiated around day 8, providing an extended duration of approximately 56 days for comprehensive evaluation of treatment efficacy.
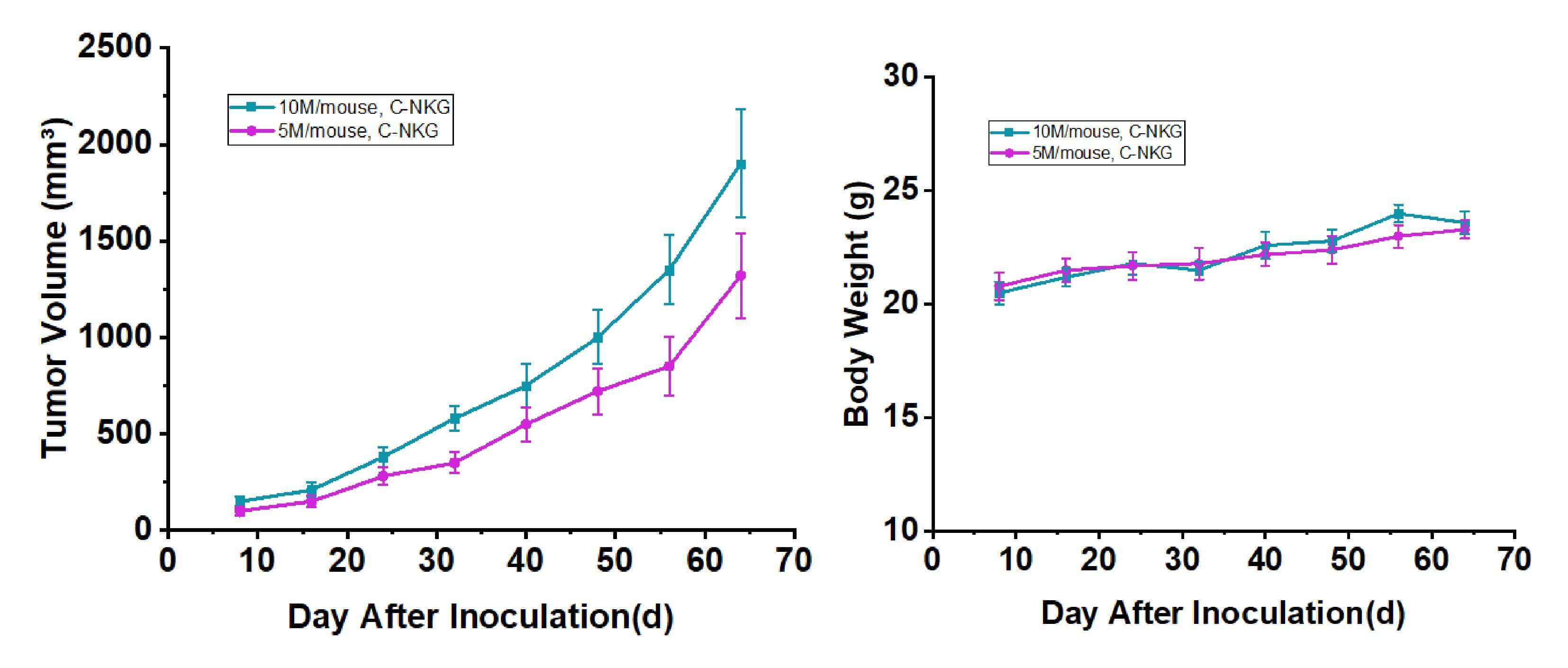 Fig. 2 Tumor volume and body weight growth curve of bladder cancer 5637 cell line subcutaneous implantation (n=6). Data are presented as mean ± standard error (SEM). (Source: Alfa Cytology)
Fig. 2 Tumor volume and body weight growth curve of bladder cancer 5637 cell line subcutaneous implantation (n=6). Data are presented as mean ± standard error (SEM). (Source: Alfa Cytology)
Contact Us
In vitro modeling services for bladder cancer provided by Alfa Cytology offer a comprehensive and cutting-edge approach to advance research and development in this field. With our expertise, state-of-the-art technologies, and commitment to scientific excellence, we aim to support researchers and pharmaceutical companies in their quest for improved diagnostics, novel therapeutics, and personalized treatment strategies for bladder cancer.
If you are interested in our in vitro modeling services, please contact us today.
Reference
- Zhu S., Zhu Z., and et al. Preclinical Models for Bladder Cancer Research. Hematol Oncol Clin North Am. 2021, 35(3): 613-632.
For research use only. Not intended for any clinical use.
Related Services


 Fig. 2 Tumor volume and body weight growth curve of bladder cancer 5637 cell line subcutaneous implantation (n=6). Data are presented as mean ± standard error (SEM). (Source: Alfa Cytology)
Fig. 2 Tumor volume and body weight growth curve of bladder cancer 5637 cell line subcutaneous implantation (n=6). Data are presented as mean ± standard error (SEM). (Source: Alfa Cytology) 